
NaCl (aq) + KCH 3COO (aq) → NaCH 3COOH + KClĪfter checking the solubility rules, we see that NaCH 3COOH and KCl are both soluble. Would a precipitate form if aqueous solutions of NaCl and KCH 3COO were mixed together? Let’s write the molecular equation: The net ionic equation can be written from the precipitate, AgCl(s), and the spectator ions from the aqueous KNO 3 (aq). In fact, the net ionic equation and the spectator ions can be determined from the molecular equation. The K + and NO 3 – ions are spectator ions. The net ionic equation shows only the reaction between the ions to form the precipitate. The potassium ions and nitrate ions cancel from both sides of the equation. The net ionic equation can be written as:Īg + (aq) + NO 3 – (aq) + K + (aq) + Cl – (aq) → AgCl (s) + K + (aq) + NO 3 – (aq) We see that AgNO 3, KCl, and KNO 3 are aqueous–they are in solution as dissolved ions. We now will write an ionic equation from our molecular equation. The balanced molecular equation is our overall chemical equation. This equation is balanced, and our molecular equation is: Add the phase labels to the products.ĪgNO 3 (aq) + KCl (aq) → AgCl (s) + KNO 3 (aq)įinally, balance the equation. AgCl is insoluble in water and is the precipitate. KNO 3 is soluble in water, therefore, it is aqueous. It is important to determine if the products are aqueous or solids. Then replace the potassium in KCl with silver. The easiest way to do this is to replace the Ag in AgNO 3 with the potassium from KCl. Both reactants are aqueous which means they are dissolved in water. Include the phase labels for the reactants. To write the chemical reaction, we include the reactants and the products. When the precipitate forms, there are NO 3 – and K + ions dissociated in the solution. You can think of the Ag + and Cl – ions as having a greater affinity for one another than for water molecules. When the two solutions are mixed together, the silver, Ag +, and chloride ions, Cl –, form a precipitate of solid AgCl. Soluble ionic compounds dissociate in water as ions. Note, the AgNO 3 and KCl are both dissolved in water and are present as ions. Two aqueous solutions, AgNO 3 and KCl are mixed together (please see figure below). These are also called double replacement reactions. Next, we look at precipitation reactions. Please get familiar with the solubility rules as they are important when writing equations. 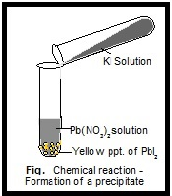
Both K 2S and CaS are soluble, but MgS and FeS is insoluble in water. Please become familiar with them as they are used in the lecture and lab quite often.įrom the table, we see that Na 3PO 4 is soluble while Ca 3(PO 4) 2 is not soluble. Below is a table of solubility rules for ionic compounds in water. In fact, we consider it insoluble in water. It is obvious that silver chloride is not very soluble in water. The solubility of AgCl, silver chloride, in water at 20 oC is 0.018 g AgCl/100 mL H 2O. Sodium chloride is readily soluble in water. The solubility of sodium chloride is 36.0 g NaCl/100 g H 2O. For example, 36 grams of sodium chloride can be dissolved in 100 grams of water at 20 oC. 
Many ionic compounds are soluble in water and others are not soluble in water. Other names for precipitation reactions include:īefore we discuss precipitation reactions, we will talk about solubility of ionic compounds in water. In this guide, we will learn how to predict the products of a precipitation reaction. A precipitation reaction is a reaction where aqueous solutions of soluble ionic compounds (salts), when mixed, produce an insoluble product called a precipitate.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
